As the throughput in the drug discovery process of large molecules have, in recent years, become considerably more high throughput many are looking for ways to automate their sample management.
In the development of recombinant proteins, plasmid DNA need continuously to be remade from source stocks to be used for transfection. For anyone looking to automate their plasmid production process, this peer-reviewed article written by Karen Billeci and colleagues at Genentech would be highly informative. Here, the authors outline how an automated process for MiniPrep was set up in their lab that purified 24,000 plasmid samples per month. The aim was to get an automated process for high throughput plasmid MiniPrep that generated purified plasmid DNA of the same or higher quality than the previously used semiautomated method. Not only is the final process described, but also the experimental work that was carried out to set up this method and ensure the quality of the final samples.
Transformation
First, automated transformation was performed on a Hamilton Microlab STAR liquid handling system that mixed plasmid DNA and competent cells. After incubation, automated colony picking was performed on Hudson RapidPick Complete system and the colonies were inoculated into deep-well plates and incubated again.
Plasmid Purification Setup
For sample preparation and plasmid purification, a customized system was designed out of three Oasis (LM900) platforms with 29 SBS positions each integrated with a robotic arm on a rail to move samples between the connected platforms and remove used consumables. One system was used for sample preparation and the other two for sample purification – the latter equipped with vacuum stations for tip drying and positions for stacked plates. Two pumps located underneath the platforms refilled on-deck reservoirs with buffers. In addition, on one end the integrated system was equipped with a hotel for plates and tip rack. This automated system for plasmid purification enables purification of up to 16 96-well plates per run.

Automated purification of plasmid DNA was performed using MiniPrep PhyTip® columns. These columns replaced the previous solution using 96-well formats of silica membranes in a semiautomated vacuum-driven chromatography method. The problem with this solution was that variations in cell culture conditions and sample pellets made the transfer of the supernatant with plasmid DNA from the processing well to the membrane difficult. This would cause dripping and required the manual control of the samples. MiniPrep PhyTip columns were selected for plasmid purification as due to the following reasons:
- -this method is able to produce sequencing and transfection grade plasmid DNA
- -this method is compatible with standard lab automation liquid handling equipment
- -this method does not require any hands-on processing
- -this method is scalable to the throughput required for this process.
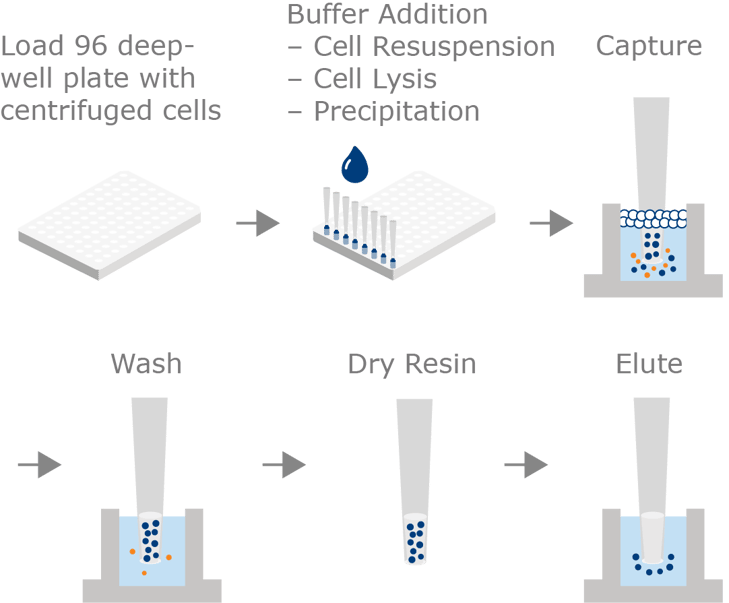
Overview of the process of Plasmid DNA purification with MiniPrep PhyTip® columns. Centrifuged cells are resuspended, lysed and precipitated in a 96 wellplate and plasmid capture on MiniPrep PhyTip columns is performed directly from the same wells by dual flow chromatography.
Quality of purified Plasmid DNA
As mentioned above - the plasmid DNA from the new automated method needed to be of the same yield and purity as had been achieved by the previously used semiautomated method. The 1 mL MiniPrep PhyTip columns with an 80 µL resin volume were selected after a comparison with the semiautomated method and MiniPrep PhyTip columns with a 200 μL tip volume. The results suggested better liquid dynamics in the larger PhyTip column with higher yield and purity as a result. The plasmid yield from automated plasmid purification by 1 mL MiniPrep PhyTip columns was around 10 μg and A260nm/A280 nm ratios >1.8. These results were very similar to the semiautomated method.
When thousands of cell samples will be purified every month there are bound to be variations in samples. Testing the robustness of an automated method allows you to foresee potential problems that may show up as a result of this. The robustness of the plasmid purification method was therefore evaluated by testing of extreme cases, for instance in cell culture density. Plasmids were purified from ideal, compact and overgrown cell cultures. The plasmid yield was the same or slightly higher for the automated method compared to the semiautomated method for ideal and compact cell cultures. For overgrown cell cultures, the yield from the automated method was significantly lower.
Transient transfection efficiency
As the purified plasmid DNA was to be used for transient transfection for expression of recombinant proteins, the transient transfection efficiency was evaluated.
Transient transfection efficiency was tested using a plasmid expressing green fluorescent protein (GFP) with a forward and reverse approach with three different transfection kits. COS7 cells were transfected with the plasmid. The transfection efficiency of the plasmid sample was determined my imaging the transfected cells and counting the number of cells that had turned green by taking up the GFP gene. In general, the transfection efficiency was comparable between the manual and automated methods.
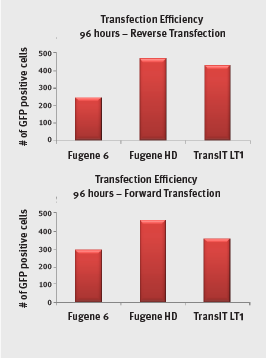
Transfection efficiency as the number of transfected COS7 cells transfected by three different transfection kits and plasmid DNA purified with the automated MiniPrep method.
More information and data about MiniPrep PhyTip® columns can be found in this whitepaper - Lysate Direct PhyTip Columns for Plasmid DNA Purification

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership