All research and development stages that involve protein assays require the essential step of protein purification. Regardless if you are working with development of a monoclonal antibody drug (mAb) or enzyme engineering, obtaining a highly pure protein sample in sufficient yield is crucial for reliable experiments. The number of samples that need to be purified may vary drastically. For example, during drug discovery, high-throughput screening of many different mutations for affinity studies require fast parallel isolation of small volumes of recombinant protein samples. Similarly, during expression screening, protein analytics are applied to many protein samples to determine which DNA construct, expression platform or other cultivation parameters lead to the expression of a biological functional homogenous protein in sufficient quantities.
Regardless of the application, good protein purification is necessary to:
Keep on reading to learn more about protein purification and about automation of this process.
Protein isolation may seem trivial, but this process may prove to be more critical for the rest of your workflow than at the first thought. To prepare the protein sample for e.g. protein crystallography for structural analysis, characterization by mass spectrometry or affinity screening by SPR, it is necessary to isolate protein from the other components present in the cell – such as other proteins, RNA, DNA and cell membrane components. Insufficient purification will cause contaminants to interfere in the assay, which may cause ambiguous results that are difficult to interpret. Reliable and reproducible protein purification is a way of ensuring the downstream assays will run smoothly with less artifacts, less false negative/positive results and with less reruns.
A convincing example highlighting the importance of protein purification was illustrated in a study encompassing the development of a telomerase activity assay. Telomerase activity assays are otherwise typically performed on crude cell lysates without prior protein purification. The study showed how the assay results were dramatically changed when a purified telomerase protein was used in the assay instead of crude samples.
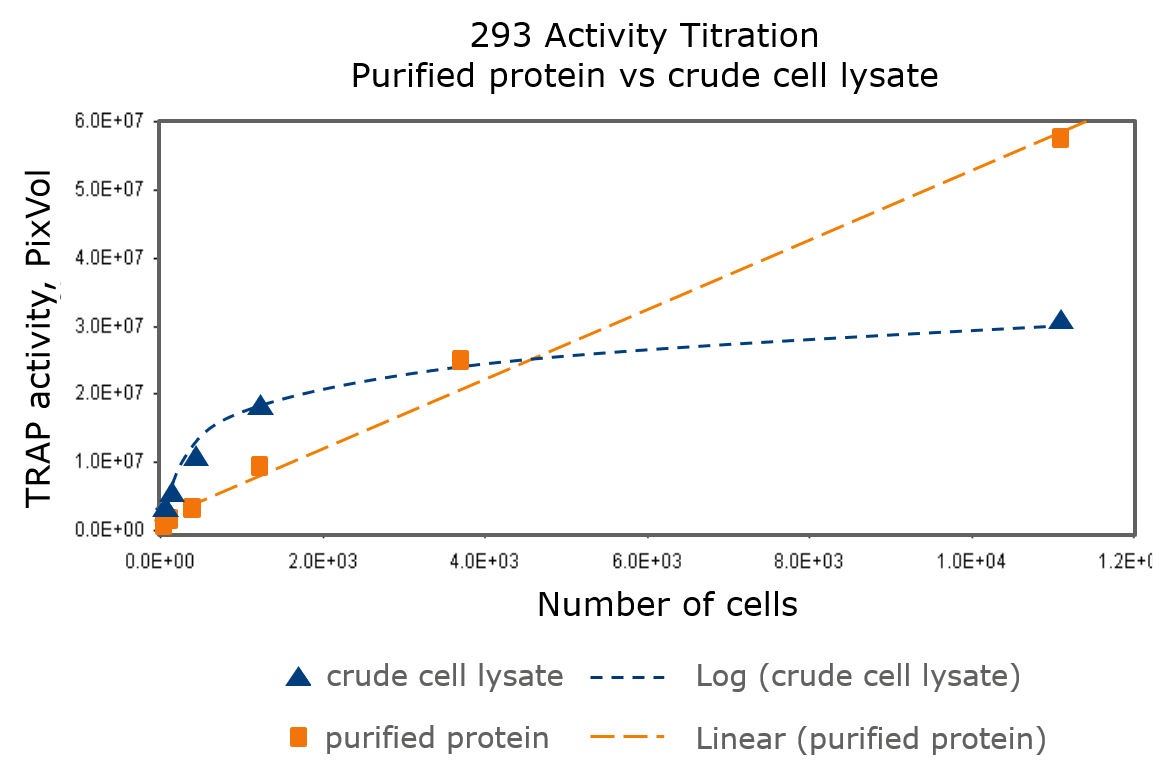
Figure 1. Comparison of Telomeric Repeat Assay Protocol (TRAP) assays performed on a dilution series of 10,000 to 100 HEK cells using 10 µg of antibody. The assay was tested on crude cell lysate or using purified protein [1].
These results indicate that there exists components beside the telomerase protein in the cell lysates that interfere with the results of the assay, resulting in a non-linear response in the assay with increasing number of cells in the assay. Meanwhile, the activity response from isolated protein gives a linear response to the number of cells in the assay, as a result from specific interactions with the telomerase protein.
Typically, laboratory-scale protein purification include the following steps: cell harvest and cell lysis (for non-secreted proteins), sample clarification and protein purification in one or several consecutive steps.
This step looks different depending on the type of cell the protein is expressed in, and where in the cell the protein will be harbored. Typical expression platforms for recombinant proteins are insect cells, bacteria cells such as E. coli, mammalian cells (mammalian cells; Chinese hamster ovary (CHO) cells and human embryonic kidney (HEK293) cells), or other eukaryotic cells.
However, for intracellular proteins or membrane proteins, cell lysis is necessary. Cell lysis can be achieved for instance chemically, osmotically, enzymatically, by bead mill homogenization or by ultra-sonication.
A sample clarification step is required to remove all insoluble material of the sample, such as cell debris so this will not clog the chromatography purification columns in the next purification step. The samples are centrifuged and/or filtered, and the protein is purified from the supernatant where the protein is solubilized.
The protein can be purified using a number of different methods such as magnetic beads, gravity columns, spin columns, PhyTip® columns or fast protein liquid chromatography (FPLC). This typically involve three different steps:
Depending on the purity requirements of the sample, more than one purification step can be used. For instance, if the sample is to be used for in vitro studies, the requirements of the sample purity will be very high and include a capture step, intermediate purification step and polishing step. For purification of histidine-tagged proteins with IMAC affinity chromatography, an additional step can be used to remove imidazole that may be used in the elution buffer. In addition, if the purification step causes the sample to become too dilute, which is a common problem, an additional step may be required to concentrate the sample by gel filtration desalting columns.
Protein purification is performed in various scales and there are a number of different technologies and protein extraction protocols to choose from when working in laboratory scale. Depending on what throughput you are working with, which protein and whether you require automation or not may influence your choice.
For protein purification by magnetic beads, polymer beads called superparamagnetic beads are used that are only magnetic when in the presence of a magnetic field. The surface of the beads are coated with various affinity ligands to which the biomolecule binds to during the capture step (immunoprecipitation). A magnet is used to separate the beads and the supernatant during the wash step. This process can be automated on a liquid handling system together with magnetic plates or rods and can also used for high throughput purification. However, the capacity of the magnetic beads can be limiting in terms of protein yield.
Affinity or chromatography resins may be packed in spin columns that are placed in microfuge tubes and eluted through centrifugation. Throughput is low and the process is primarily manual, but easy to use and well known in the research community.
In FPLC, affinity media or chromatography resin is packed in typically a 1 mL or 5 mL column, connected to a liquid chromatography system and run at medium pressure. Throughput is relatively low, although new systems exist that enables parallel processing of several samples at the same time.
Small resin bed of e.g. affinity chromatography media packed in the tip of a pipette tip between two inert screens for automated protein purification by liquid handling systems, such as Hamilton Microlab® STAR™, Tecan Freedom EVO® or MEA 2. Sample capture, wash and elution is performed by pumping the liquid back and forth over the packed resin bed (a process called dual flow chromatography [2]). Besides automation, this process comes with some added advantages to the sample, such as high recovery and reproducibility.
The process can be automated on a liquid handling system and used for low to high throughput protein purification.
Figure 2. PhyTip columns can be used on virtually any liquid handling system for automated protein purification.
Blog post: Dual Flow Chromatography - Protein Purification in a Pipette Tip
In batch binding, the protein sample is mixed with a resin of affinity media for specific binding of the protein to the resin. The mixture is then filtered for washing to remove contaminants and elution of the protein from the resin. This process typically takes much longer to achieve the same interaction of the protein sample with the resin.
For the use of gravity columns for protein purification, a column is packed with resin and the sample is applied to the top of the column and flows through the column by gravity flow. This is typically a manual process, and typically require the most hands-on time.

Figure 3. A gravity column. The sample is added to the top of the column manually.
Blog post: Technologies for Automated High-throughput protein purification.
In affinity chromatography, the specific interaction between the protein and a ligand with high binding affinity to the protein in question is used. The ligand is immobilized to a chromatography resin, such as highly cross-linked agarose (sepharose).
Antibodies consist of two heavy chains and two light chains joined together by disulfide bonds. Both the heavy chain and the light chain have constant regions and a variable region at one end. The antigen binding sites constitute the variable regions of both the heavy and the light chains. The light chains exist in two different types: kappa and lambda.
The most common affinity ligand in antibody purification by far is Protein A, which binds to the Fc region of IgG antibodies. Protein A and Protein G are proteins that are naturally produced by bacteria. However, modern affinity media typically contain recombinant engineered versions of this protein for improved binding characteristics – one example is MabSelect™ Sure™ (GE Healthcare). During affinity purification with protein A and Protein G media, the elution buffer has a low pH. Consequently, the protein a purification protocol also includes a neutralization step after elution.
Recombinant protein technologies enables scientists to develop biopharmaceuticals in the form of antibody fragments as well, which contain only certain parts of the antibody structure including the antigen-binding region. For antibody fragments lacking the Fc-region, other affinity media are required for purification, typically with specificity to the kappa or lambda regions of the light chains. One example is protein L that bind to the variable region of the kappa light chain.
Recombinant antibodies or other recombinant proteins can be expressed carrying an affinity tag that is used for purification. The purification tag may influence the protein’s solubility, stability and biological activity.
The histidine tag, typically consisting of six histidine amino acids is by far the most common tag used for protein purification. For purification of his-tagged protein, immobilized metal affinity chromatography (IMAC) media is used, charged with either Nickel (Ni-IMAC) or Cobalt (Talon).
The binding interaction between streptavidin and biotin is the strongest non-covalent interaction that exists with a KD of 10-15 M. This can of course also be used for protein purification of biotinylated proteins that are purified with affinity media with immobilized recombinant streptavidin-ligands. Other examples are the GST-tag and the DDDDK-tag. Proteins with the GST-tag are purified using an affinity media with immobilized Glutathione protein that bind to the GST-tag. The DDDDK-tag can be used for very gentle protein purification by the use of a media with immobilized anti-DDDDK antibodies.
Reversed phase chromatography can be effectively be used for purification of protein or peptide samples to remove salts before e.g. mass spectrometry analysis. Because the elution buffer from a revered phase purification will denature the protein, this is not a suitable method when protein activity is required.
Ion exchange resins exist for protein purification as weak/strong anion or cation and can be used for purification of other biomolecules. Molecules are separated depending on their net surface charge, influenced by the isoelectric point and buffer pH. The protein is eluted with increasing ionic strength with ions competing for the binding sites, or change in pH to change the net charge of the protein.
Size exclusion chromatography (SEC) resins contain beads, with pores of various sizes and can be used for protein purification and purification of other biomolecules. Molecules are separated in terms of size, as smaller molecules will be retained longer on the column due to longer migration path through the column. Consequently, in SEC the proteins do not bind to the resins such as in for instance affinity chromatography, but rather sieved through the resin pores. As a result, the purification is not affected by buffer pH or other components. Desalting gel filtration columns remove small molecules effectively and are also used for buffer exchange and sample desalting.
Low recovery during protein purification is a common problem. Recoveries down to 30 % is not unusual. This is typically due to fast flow through the purification column during the capture step, so the protein is not given sufficient time for binding to the stationary phase. As a result, much for the protein will pass through the column without binding to the media.
Dilution of the protein sample during the protein purification process is a common problem and is a result of the large elution volumes required during the elution step. Dilution of the sample and consequently low concentrations of the protein in the purified sample can be problematic in following assays. An additional step may be required to concentrate the sample after purification.
Low repeatability of protein purification results is typically due to inconsistent flow through volumes during e.g. elution from chromatography columns, which can be the case when vacuum or centrifugation is used for this step. Low reproducibility and inconsistent results from the protein purification process can lead to problems if you attempt to streamline your entire recombinant protein expression workflow and want to handle all protein samples in the same way. Inconsistency increases the need of re-runs. Low reproducibility in purification results makes it more difficult to set up processes for the following steps after the purification in your workflow.
Something many of us know all too well is that protein purification can be associated with repetitive manual labor in the lab – especially when protein purification requires more than one purification step and there is a high number of samples. Doing the same thing repeatedly is not too inspiring, and the risk of errors increase when we are bored. Another problem is repetitive strain injury that affect too many laboratory technicians due to repetitive pipetting. A Swedish study in 1994 found that laboratory technicians had an increased risk of hand and shoulder ailments, directly related to the extent of manual pipetting they were undertaking. The impact was so large that they posted a suggestion for permissible exposure levels of pipetting per year [3].
As it is essential to maintain the biological activity of proteins such as enzymes during the purification process, gentle purification processes are required for following biological assays. Along with low sample recoveries, protein inactivation through aggregation or denaturation during purification is a common problem due to harsh purification conditions. For instance, one-pass chromatography leads to a concentrating effect of the protein sample in the beginning of the columns, which can result in some of the protein sample to aggregate.
If you are dealing with an increasing number of protein purification samples and need to standardize your workflow, lab automation may be a solution to improve productivity and streamline your protein expression and purification platform.
In general, process automation provides higher reproducibility than manual work. An interesting example of how the variations between samples are smaller after automated sample purification compared to manual sample purification is shown in Figure 4. When it comes to protein purification, automated sample purification by PhyTip columns typically give a CV value of 3-5%.
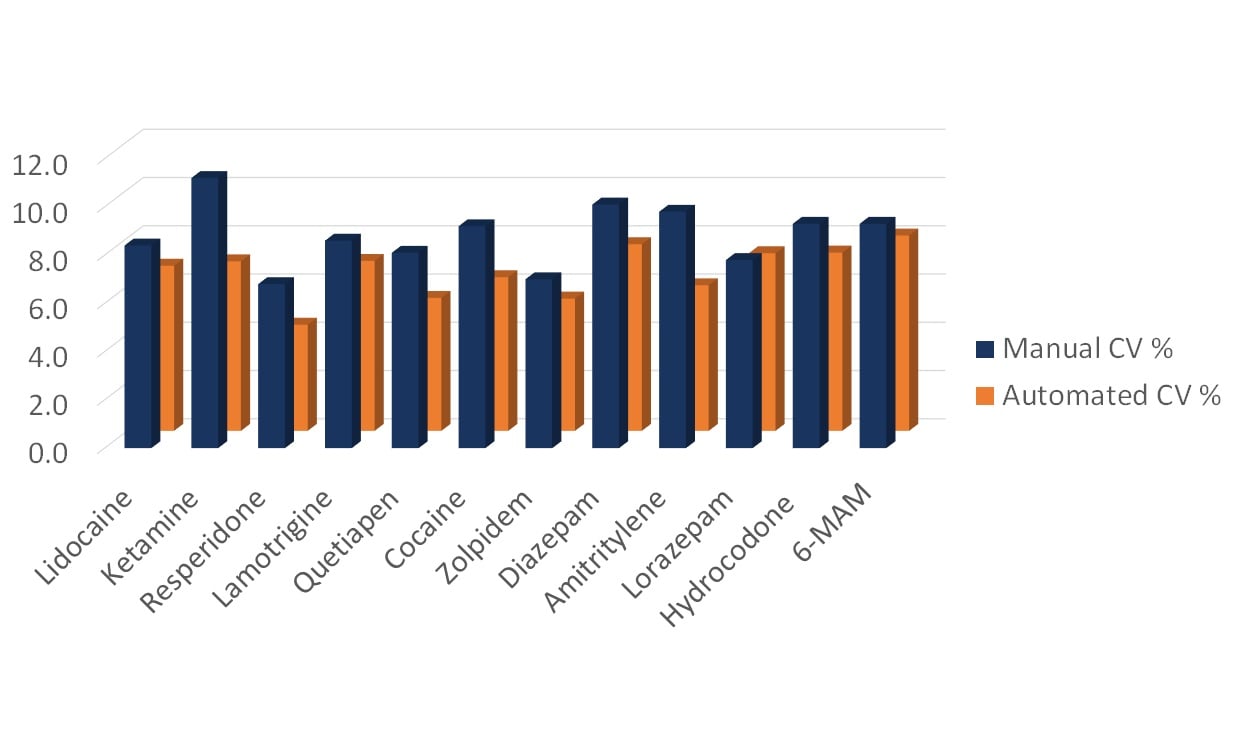
Figure 4. Variations in responses for 12 different compounds as a result of manual vs automated extraction from blood for forensic analysis [4].
When it comes to repetitive manual work, it may not come as a surprise that automated systems perform better than laboratory personnel. We all make mistakes – and boredom from doing the same thing repeatedly makes us more prone to making mistakes.
Ergonomic-related injuries are far too common for laboratory personnel, due to repetitive manual work such as pipetting. By allowing a robotic system to take over the repetitive motions, these injuries can be prevented and enable workers to focus on more high-level tasks that they do best. If you are working with hazardous compounds or liquids, automation will also reduce exposure.
Purchasing a new liquid handling robot may seem like a big investment with a price tag between 50,000 and 150,000 USD. It should however be taken into account that is enables highly educated staff can make more effective use of their time for other work tasks.
During the last years, the biopharma industry has considerably started to screen a higher throughput of protein samples, which put higher demands on effective protein expression workflows. Introducing automation is a great way of improving your productivity in your lab and remove the potential bottleneck that protein purification may cause. Automated liquid handling system are able to work both during weekends and nights, so it enables you to even get some work done during off hours.
Choose a solution that works for your throughput. Liquid handling systems with 96 channel heads enable high throughput sample processing but comes with a price tag of 100,000 – 150,000 USD, so you may want to wait getting one until when your throughput demands it. There are also several smaller to mid-throughput solutions that are more economical and are easier to operate. However, do take into account that your throughput may increase with time, so a scalable automated solution can be a good idea.
Also, choose a solution that can be applied to every protein you are working with
There are both customized and off-the-shelf solutions for laboratory automation. Whereas off-the-shelf solution often leaves very little flexibility in terms of labware and how you treat your samples before and after the solution, a customized system enables you to get a solution that is exactly according to your requests. However, after the installation and validation of your customized solution is finished, implementing new changes to it will be more laborious that you may expect (for instance changing the labware).
Workflow automation can be tricky, and implementation of a new application can be an investment in both time and money. Choose a vendor with excellent customer support to make sure your solution will really work for your application, as it may be difficult to program a new protocol by yourself.
Protein purification is an essential part in research and development of new proteins. Generation of highly pure protein samples every time leads to less false positives or false negatives in the consecutive assays where the protein is analysed. In short - this step must always work to avoid potential problems in the following steps. Automation can be a great way to ensure consistent protein purification results, do labor savings and improve your productivity. Choosing the automation solution that is right for your can however be challenging. Make sure to choose a solution that suits your throughput and overall workflow and a vendor that is highly experienced in automated protein purification.
Interested in learning more? Download our whitepaper - PhyTip® Columns Processed on the Tecan Freedom EVO® for Complete Automation of High-Throughput Protein Purification and Sample Preparation

Guenther B, D Gjerde (2017) Dual Flow Chromatography, LCGC Europe Vol 30, Issue 8, pg 404–411
Björksten MG, Almby B, Jansson ES (1994) Hand and shoulder ailments among laboratory technicians using modern plunger-operated pipettes. Appl Ergon, 25(2): 88-94'
Youssef M. et al., (2019). Low Cost Entry for Automation in Laboratory Settings. Poster P211 presented at the Society of Forensic Toxicology (SOFT), San Antonio, TX, USA.
Biotage is a Global Impact Tech Company committed to solving society’s problems. We offer workflow solutions and products to customers in Drug Discovery and Development, Analytical Testing and Water and Environmental Testing.