Immunoprecipitation is a well-known method in biomedical research for protein isolation. This method is useful to be able to study a protein’s physiochemical properties, post-translational modifications or expressions in various tissues or cell lines. In immunoprecipitation, the protein is captured on a solid support with an immobilized antibody that binds to the protein in question.
Immunoprecipitation is usually carried out in small tubes by the use of magnetic beads or loose affinity resin as solid support.
It is also possible to do small-scale immunoprecipitation using affinity columns packed in a pipette tip. This method is called High Performance Immunoprecipitation (HPIP) and uses an electronic pipette to pump the sample back and forth across the resin bed for full capture of the entire sample (a process called dual flow chromatography). The advantages of this method is that it fast, easy to set up, provides highly pure samples and that it is automatable.
How does it work?
Each Protein G PhyTip® column, is a Protein G affinity column packed between two thin screens in the tip of a pipette tip. Alternatively, a Protein A PhyTip® column (or any other affinity media with affinity to the Fc region) can be used. The antibody is immobilized on to the affinity support as Protein G binds to the Fc region of the antibody, and the protein binds to the antigen binding site of the immobilized antibody.
Direct vs. Indirect IP
Immunoprecipitation can be done in two ways - direct or indirect . In direct immunoprecipitation, the bridging antibody is first added to the Protein G affinity media and allowed to bind to it. In the second step, the immobilized bridging antibody is used for purification of the protein of interest from the sample.
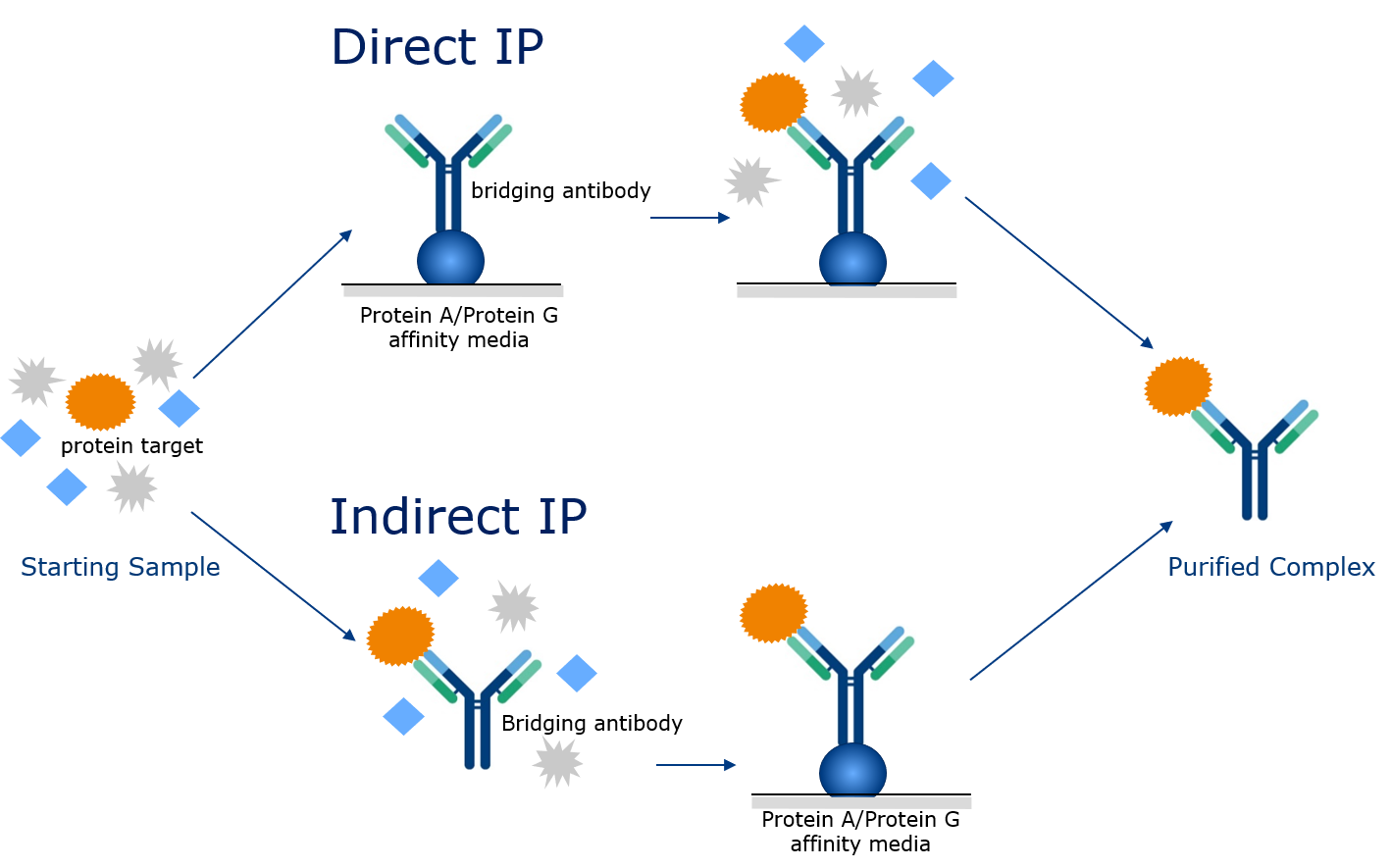
So, when do you use direct IP and when should you use indirect IP? Direct IP is preferred when the binding kinetics of antigen protein to antibody is fast. Indirect IP is preferred when the binding kinetics of antigen protein and antibody is slow.With indirect immunoprecipitation, the bridging antibody is premixed with the sample containing the protein, and the protein of interest binds to the bridging antibody. The sample mixture is then purified, by the use of Protein A or Protein G affinity media, and the bridging antibody-protein conjugate is captured on the affinity media.
Setting up a High Performance Immunoprecipitation method in your lab
Two examples of the use of Indirect HPIP and Direct HPIP can be found in these two application notes (listed below). Here, HPIP was used for purification of a GST-tagged protein by the use of Protein G PhyTip columns and a bridging antibody with binding affinity to the GST tag.
For these experiments, a hand-held electronic pipette was used to pump the samples back and forth across the packed resin bed with a controlled flow for a semi-automated protocol - meaning that the user moves the pipette between the various purification steps and place it in the correct wells. For full automation, the same protocol can easily be transferred to a liquid handling system (such as Tecan Freedom EVO®, Hamilton STAR™, the MEA 2 system etc.).The bridging antibody was captured on the Protein G resin by slowly pumping the sample back and forth across the packed resin. Both the direct and indirect methods produced replicable protein bands with high intensity on SDS-PAGE gels due to highly concentrated purified protein samples, very low background from contaminants and relatively fast processing of samples.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership